Risperdal
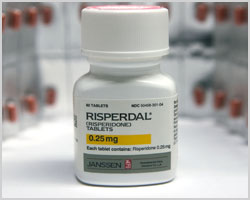 Ripserdal, the trade name for the anti-psychotic drug Risperidone, was approved for use in the treatment of schizophrenia and bipolar disorders by the U.S. Food and Drug Administration in 1993. It has also been recommended for certain “off-label” or unapproved uses, including psychosis, aggression and behavioral challenges in elder patients with dementia.
Ripserdal, the trade name for the anti-psychotic drug Risperidone, was approved for use in the treatment of schizophrenia and bipolar disorders by the U.S. Food and Drug Administration in 1993. It has also been recommended for certain “off-label” or unapproved uses, including psychosis, aggression and behavioral challenges in elder patients with dementia.
In 2015, the FDA notified Janssen-Cilag, Risperdal’s manufacturer, that it must include what is known as a “black-box warning” on Risperdal labels, recommending that the product not be used to for treat seniors with dementia.
Since its introduction into the American market, Risperdal has been linked to a wide range of side effects, from blurred vision, dizziness or drowsiness and headaches to lethargy, indigestion and weight gain. Other side effects tied to use of Risperdal include:
- Blood clots
- Chest pain or irregular heartbeat
- Stroke or transient ischemic attack
- A dramatic change in blood pressure
- Abnormal liver function
- Bowel obstructions
- Atrial fibrillation
If you or someone you love has suffered injury using the Essure birth control product, GetLegal has the tools and resources to help you find skilled legal representation to protect your rights. Visit our attorney directory, look for your geographic location and find the best lawyer for your situation.
Connect with Top-rated Attorneys Near You
Sponsored Advertisement
Defective Drugs & Medical Devices Topics
Accutane
Aciphex
Actonel
Actos
Adderall and Ritalin
Advair
Aldara
Alendronate
Alli
Ambien
Amiodarone
Anzemet
Aptivus
Aranesp
Arava
Aredia
Aricept
Avandia
Avastin
Avelox
Avodart
Avonex
Azathioprine
Benicar
Clozaril
Concerta
Coreg
Crestor
Cylert
Cymbalta
Cytotec
Darvocet
Darvon
Daypro
Essure Birth Control Medication
Gadolinium
Granuflo
Hydroxycut
Invokana
IVC Filters
Ketek
Levaquin
Mirapex
Neurontin
Onglyza
OxyContin
OxyContin Birth Defects
Paxil
Power Morcellators
Pradaxa
Propecia
Reglan
Risperdal
Surgical Mesh
Tequin
Trasylol
Viagra
Xarelto
Xolair
Zelnorm
Zoloft
Exactech Hip and Knee Replacement Devices Subject to Recall
Latest Article
Joint Custody vs. Sole Custody: What Courts Prefer Today
Custody decisions are not about rewarding or punishing parents. Courts focus on one question: what arrangement best supp... Read More
Parenting Plans: What Must Be Included for Court Approval
Courts don’t approve parenting plans because they look fair to parents. They approve them because the plan clearly pro... Read More
Employee Misclassification: Independent Contractor vs. Employee
How a worker is classified matters more than many people realize. Being labeled an independent contractor instead of an ... Read More
GETLEGAL®ATTORNEY DIRECTORY
Find Leading Attorneys in Your Area
NEED PROFESSIONAL HELP?
Talk to an Attorney
How It Works
- Briefly tell us about your case
- Provide your contact information
- Choose attorneys to contact you